Only logged in customers who have purchased this product may leave a review.
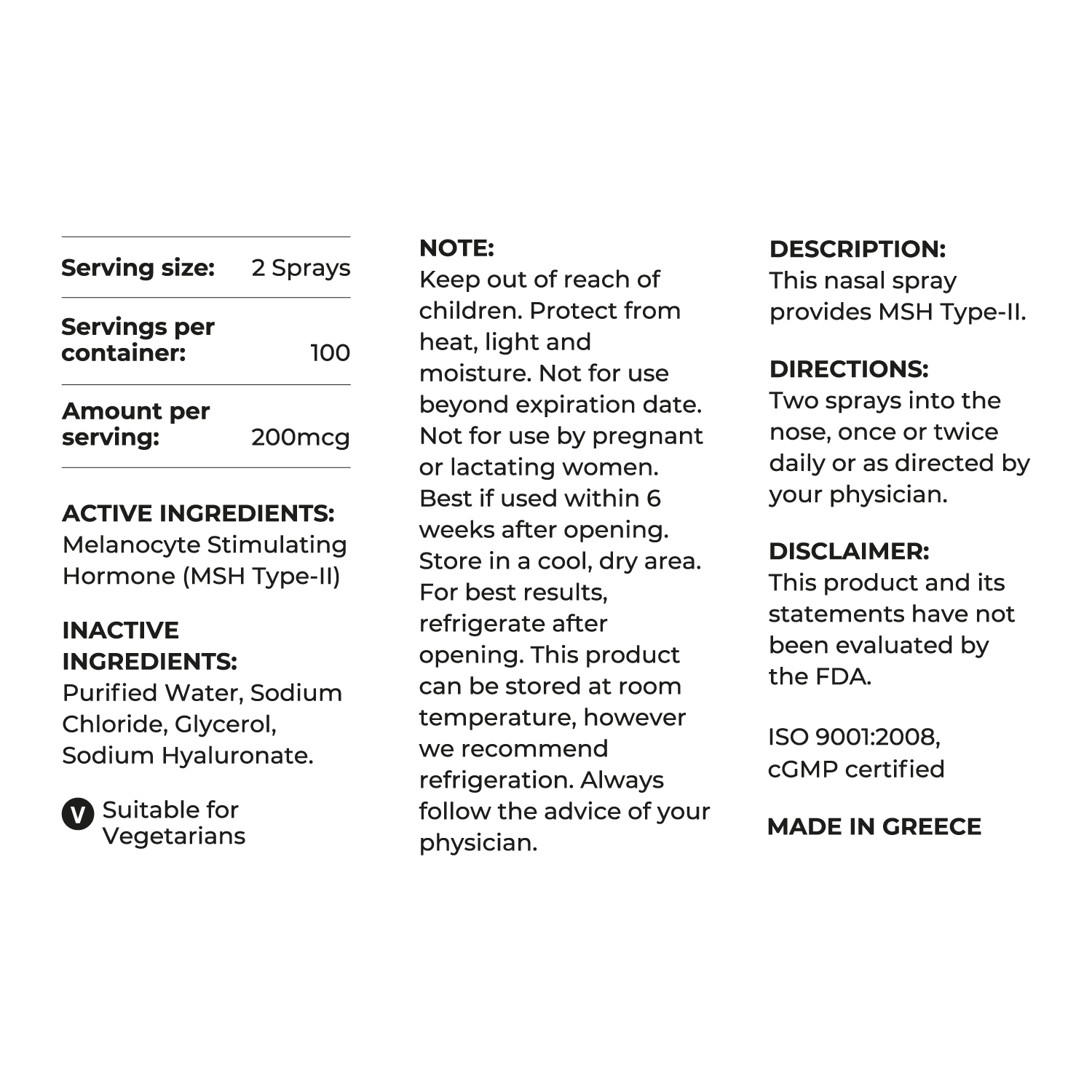
MSH2Pro™ (MSH-2)
$59.99
20ml/20mg Nasal Spray
Just one or two sprays daily help produce a natural all over tan, without the need to expose the skin to direct sunlight or to have injections.
MSH2 Also:
- Increases libido
- Works synergistically with the other hormones to rejuvenate cells and organs
Contains melanocyte stimulating hormone, that is directly involved in helping to tan the skin.
The last couple of years have seen significant disruption and rises in supply chain and distribution costs. Therefore, we have had to introduce a minimum order value of $120 (incl. shipping). We hope you understand this predicament and we remain committed to providing high quality, hard to obtain specialist health products.
In stock




Reviews
There are no reviews yet.